
Rotational device that enables multidirectional tweezer measurements at high forces
Read moreMeasuring the contractile forces of cells to the underlying substrate
Read moreTechnique to estimate the migration speed of different cell lines on 2D substrates
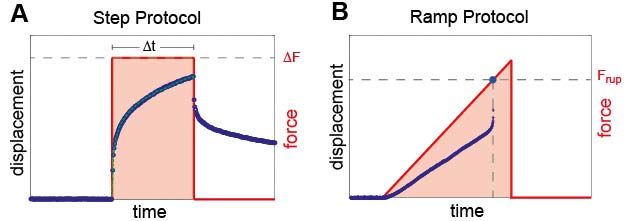
Read moreThe high force magnetic tweezer device is a scientific rheometer used to study the mechanical properties of biological materials in the micrometer range, especially the cytoskeleton of cells. Especially according to cells it is necessary to understand the dynamics of cellular force responses in order to understand cell growth and differentiation. The high force magnetic tweezer setup is based on the setup designed by Kollmannsberger et. al. 2007, which is used to estimate the viscoelastic cell properties both in the linear and non-linear deformation regime. This noninvasic technique consists of an electro magnet applying precise forces without any contact to small micron sized magnetic particles that are bound via focal adhesion complex to the cytoskeleton of cells. Using optical bead tracking during force application the displacement of the magnetic particles can be measured in real time with a precision of one thousandth of the particle diameter.
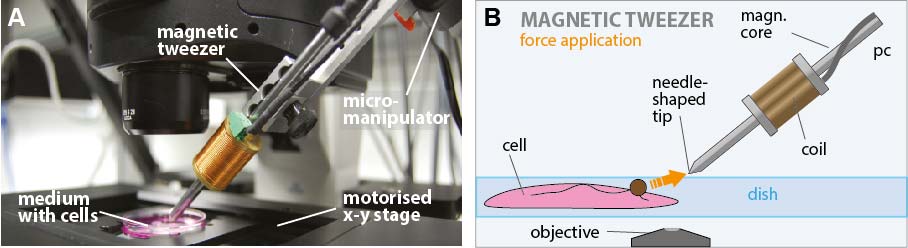
The principal item of the magnetic tweezer device is the magnetic tweezer arm shown in figure 2. It consists of a special steel bar with a diameter of 4.5 mm where the end closest to the specimen is tapered to concentrate the flux so that a large field and field gradient is created. The magnetic field itself is generated through a cooper coil wound around a brass body connected to the steel core. The magnetic field is measured by a hall probe sitting at the other end of the steel core.

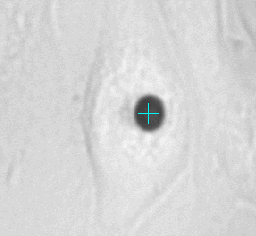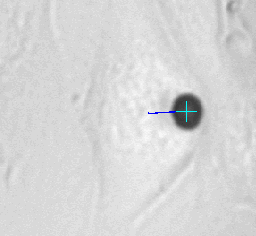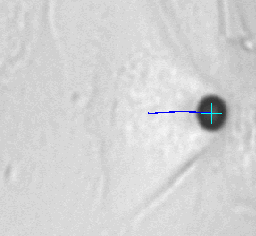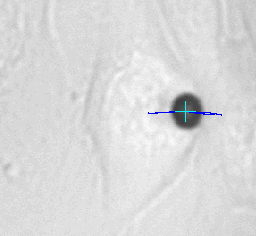
Left animation loop shows the strong deformation of a cell due to an applied force ramp up to 80nN with the high force magnetic tweezer device. This particular cell could withstand this force without bead disruption. The bead trajectory is tracked in real time. Forces of up to over 200nN can be used to investigate the adhesion strength of cells to the extra cellular matrix. Disruption forces for each cell are detected and used for further analysis.
The complete tweezer setup is shown in figure 4. The setup was maximally user friendly designed. During the whole measurement, the user has all controls in striking distance and can thus concentrate exclusively on the measurement itself. From his place all necessary adjustments can be performed from adjusting the intensity, probe positioning as well as the tweezer positioning. A wireless numpad is adjusted to the micromanipulator containing all necessary software commands needed for the tweezer software. A wireless mouse pad and keyboard enables the user to control any other adjustments on the computer.
